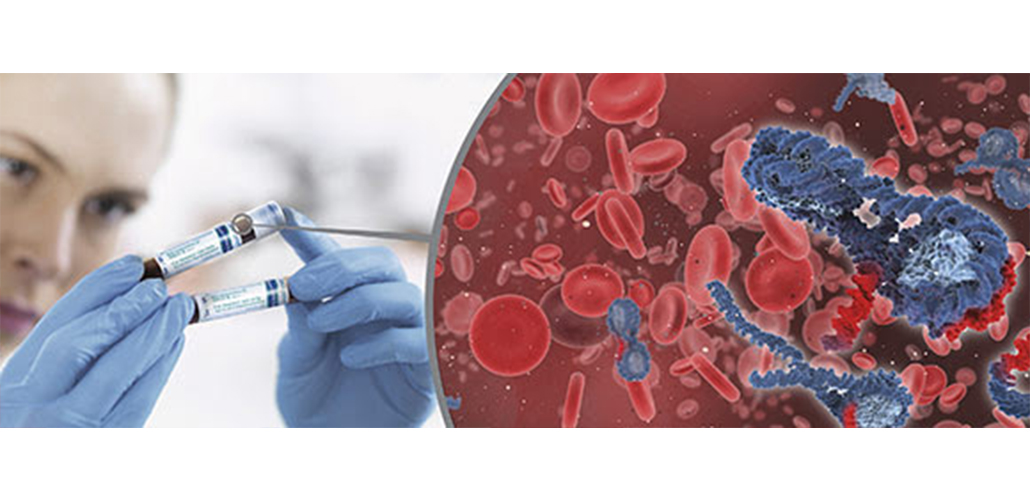
From blood collection to ccfDNA purification – standardize the process
April 05, 2017With the newly released QIAsymphony PAXgene Blood ccfDNA Kit from Qiagen, as part of the PAXgene Blood ccfDNA System, you can now standardize and streamline your circulating cell-free DNA (ccfDNA) workflow with the highest reproducibility and efficiency.
Starting from stabilization during blood collection, transport and storage to fully automated ccfDNA purification – the process helps lend consistency, high quality and high purity to your samples!

GWYAMOFG1nbnHPRLPnXn.jpg)

